Radiologia Brasileira - Publicação Científica Oficial do Colégio Brasileiro de Radiologia
AMB - Associação Médica Brasileira CNA - Comissão Nacional de Acreditação
 Vol. 54 nº 4 - July / Aug. of 2021
Vol. 54 nº 4 - July / Aug. of 2021
|
ORIGINAL ARTICLES
|
|
Correlation between 99mTc-TRODAT-1 SPECT and 18F-FDOPA PET in patients with Parkinson's disease: a pilot study |
|
|
Autho(rs): Julieta E. Arena1,a; Leandro Urrutia1,b; Germán Falasco1,c; Magdalena Ponce de Leon1,d; Silvia Vazquez1,e; Malco Rossi1,f; Marcelo Merello1,2,g |
|
|
Keywords: Parkinson disease/diagnostic imaging; Parkinsonian disorders/diagnostic imaging; Tomography, emission-computed, single-photon/methods; Positron-emission tomography/methods; Tropanes/pharmacokinetics; Dihydroxyphenylalanine/analogs & derivatives. |
|
|
Abstract: INTRODUCTION
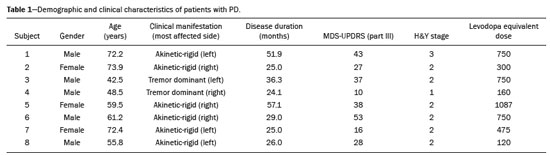
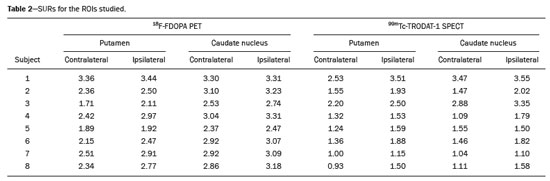
The main neurochemical hallmark of Parkinsons disease (PD) is insufficient dopamine production by the substantia nigra pars compacta. Although PD is still diagnosed on the basis of clinical findings(1), it is difficult to make the early differential diagnosis between true PD and other, nondegenerative forms of parkinsonism, such as those that are drug-induced, vascular, or even psychogenic, in which imaging studies could contribute to a more precise diagnosis(2). Several molecular imaging techniques capable of illustrating the different stages of dopamine synthesis, release, and uptake are currently available. Fluorine-18-labeled fluorodopa positron-emission tomography (18F-FDOPA PET) and dopamine transporter single-photon emission computed tomography (DAT-SPECT) have both been used for some time now, with well-established indications and diagnostic yields(3,4). Although PET provides higher spatial resolution than does SPECT and is better for quantification purposes, SPECT is more affordable and more readily available in clinical practice as well as for epidemiological studies(5,6). The use of DAT-SPECT with 123Iioflupane (DaTscan) has been approved by the US Food and Drug Administration (FDA) and European Medicines Agency (EMEA). In contrast, technetium-99m-labeled tropane derivative SPECT (99mTc-TRODAT-1 SPECT) has not been approved for use in the United States or Europe, despite the fact that 99mTc-TRODAT-1 has an effective half-life for imaging of 6.02 h and that 99mTc-TRODAT-1 SPECT is extensively used in a number of other countries(79). In addition, 99mTc-TRODAT-1 is a biomarker of the DAT, which is found on the presynaptic membrane of dopaminergic projections and plays a role in extracellular dopamine regulation(10). Several small studies have shown 99mTc-TRODAT-1 SPECT to be a more affordable, accessible, kit-based imaging alternative that can effectively distinguish between normal and abnormal subjects(11,12). The aim of this study was to investigate the correlation between 99mTc-TRODAT-1 SPECT and 18F-FDOPA PET. MATERIALS AND METHODS Subjects Eight patients with a clinical diagnosis of PD, according to current criteria(1), were recruited prospectively and randomly from among patients under treatment at a movement disorders clinic. To reduce the risk of misdiagnosis, we recruited only patients who had been diagnosed at least 2 years prior and had shown a sustained response to dopamine replacement therapy. All of the patients underwent clinical examination by an experienced neurologist and scored according to the Hoehn and Yahr (H&Y) scale(13) and the Movement Disorder Society-sponsored revision of the Unified Parkinsons Disease Rating Scale (MDS-UPDRS) part III(14) in the off state. Each subject underwent a 99mTc-TRODAT-1 SPECT and an 18F-FDOPA PET, in random order. The study protocol was approved by the local research ethics committee. All participating patients gave written informed consent. Imaging analysis Patients were required to discontinue all dopaminergic medication overnight prior to both studies. For the 99mTc-TRODAT-1 SPECT study, other potential test-altering medications, such as certain antidepressants, were also discontinued. For the 18F-FDOPA PET examination, patients were premedicated with 200 mg of carbidopa (to inhibit peripheral L-dopa metabolism) 60 min before the administration of 370 MBq of 18F-FDOPA (ANMAT certificate no. 58050; Tecnonuclear SA, Buenos Aires, Argentina). At 120 min after tracer administration, images were acquired in a 64-slice PET scanner (Discovery 690; GE Healthcare, Waukesha, WI, USA). Patients were awake, in the supine position, with their head immobilized. Images were acquired with a static (30 min) protocol and were reconstructed using the ordered-subset expectation-maximization algorithm (2 iterations; 24 subsets). For the 99mTc-TRODAT-1 SPECT examination, patients were injected with approximately 20 mCi of isotope DOPA-TEC (ANMAT certificate no. 57706; Tecnonuclear SA) and images were acquired 240 min later. Visual and semiquantitative analysis was performed for 99mTc-TRODAT-1 SPECT and 18F-FDOPA PET. Images were reconstructed by back-projection with a Butterworth filter at a cutoff frequency of 0.45, and the Chang method was employed for attenuation correction(15). For each pair of images from an individual patient, spatial co-registration was applied. The 18F-FDOPA PET images were spatially normalized to a reference 18F-FDOPA template(16). Calculated transformation was applied to 18F-FDOPA PET and 99mTc-TRODAT-1 SPECT images. Regions of interest (ROIs) for the striatum (caudate nucleus and putamen) were delineated in the 18F-FDOPA template and applied automatically to the 18F-FDOPA PET and 99mTc-TRODAT-1 SPECT images. The ROIs in the occipital cortex were delineated and applied in a similar fashion to serve as reference areas. Specific uptake ratios (SURs) were calculated for each ROI and divided by the mean count in the occipital area, the SUR for each region thus being obtained. Asymmetry indices were calculated for the caudate nucleus and putamen through subtraction of the SUR on each side, divided by the mean values for both. The readers were blinded to the clinical data as well to previous individual PET or SPECT study results. Statistical analysis The SURs obtained for the ROIs on 99mTc-TRODAT-1 SPECT were correlated with those obtained on 18F-FDOPA PET. To compensate for the signal effects due to individual variation in specific uptake incorporation, we calculated the mean signal for each ROI pair (ipsilateral and contralateral to the most affected side). To evaluate the consistency between the two acquisition methods in terms of the difference between the ipsilateral and contralateral signals, linear regression was performed and Pearsons correlation coefficient was calculated. RESULTS The sample included three women and five men. The mean age was 58.3 ± 14.3 years (range, 35.874.9 years), and the mean disease duration was 34.3 ± 14.1 months (range, 15.157.1 months). One patient was classified as H&Y stage 1, whereas six were classified as H&Y stage 2 and one was classified as H&Y stage 3. The mean MDS-UPDRS part III (motor examination) score was 31.5 ± 14.2 (range, 10.053.0). All patients had asymmetric symptoms; one (subject 5) presented mild dyskinesias and motor fluctuations (Table 1). The mean time elapsed between the 18F-FDOPA PET and 99mTc-TRODAT-1 SPECT examinations (or vice versa) was 3.6 ± 2.8 months (range, 0.48.3 months), and no treatment modifications were made during the interim. For each patient, the visual comparison between the two methods was performed by two experienced observers and was concordant across all studies. Greater striatal signal loss was observed contralateral to the more affected side (Figure 1). As expected, the caudate nucleus and putamen were both better defined spatially and more readily identifiable on 18F-FDOPA PET images than on 99mTc-TRODAT-1 SPECT images. 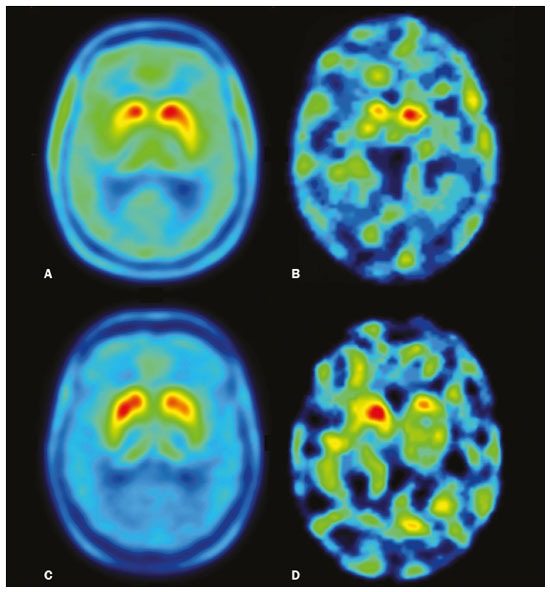 Figure 1. Representative transverse 18F-FDOPA PET image (left) and concordant 99mTc-TRODAT-1 SPECT image (right) of patients with PD (A,B: subject 7; C,D: subject 6). A,B: Note the markedly low tracer uptake in the right putamen, the uptake also being low, to a lesser degree, in the left putamen. C,D: Inversely, tracer uptake was markedly low in the left putamen and low, to a lesser degree, in the right putamen. Figure 1. Representative transverse 18F-FDOPA PET image (left) and concordant 99mTc-TRODAT-1 SPECT image (right) of patients with PD (A,B: subject 7; C,D: subject 6). A,B: Note the markedly low tracer uptake in the right putamen, the uptake also being low, to a lesser degree, in the left putamen. C,D: Inversely, tracer uptake was markedly low in the left putamen and low, to a lesser degree, in the right putamen.Semiquantitative analysis of the caudate nucleus and putamen ROIs showed greater loss of dopaminergic activity in the striatum on the contralateral side, and the degree of loss was comparable between the two techniques; except in subject 5, in whom, on 99mTc-TRODAT-1 SPECT, the ipsilateral caudate nucleus was more affected, although the degree of asymmetry was only 3.4%, which was not significant (Table 2). Taking all of the studies together, the semiquantitative analysis showed a statistically significant correlation between the two methods (r = 0.73; p < 0.01). The linear regression of predetermined ROIs (for the caudate nucleus and putamen, on the most affected and contralateral sides) is shown in Figure 2. The box plots in Figure 3 show that the differences between the ipsilateral and contralateral sides were greater on 99mTc-TRODAT-1 SPECT images than on 18F-FDOPA PET images. The results of the visual and semiquantitative analyses were concordant across all studies, as well as being consistent with the clinical findings. 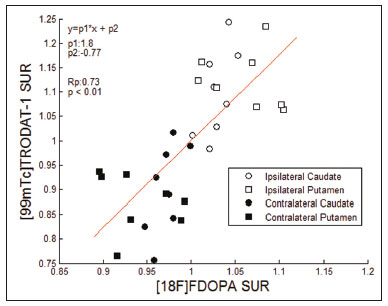 Figure 2. Correlation and linear regression of 99mTc-TRODAT-1 and 18F-FDOPA uptake in ipsilateral and contralateral ROIs. Single points represent standardized SURs for each individual putamen and caudate nucleus. 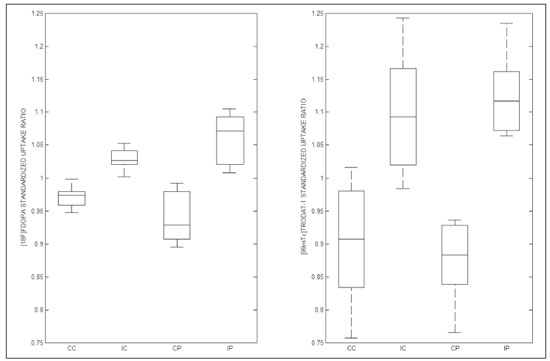 Figure 3. Box plot of standardized SUR group distribution for 18F-FDOPA (left) and 99mTc-TRODAT-1 (right). CC: contralateral caudate nucleus; IC: ipsilateral caudate nucleus; CP: contralateral putamen; IP: ipsilateral putamen. DISCUSSION In our sample of patients with mild to moderate PD, randomly selected from among patients under treatment at a movement disorders clinic, we found that 99mTc-TRODAT-1 SPECT and 18F-FDOPA PET correlated well in relation to the visual and semiquantitative analyses. The 18F-FDOPA PET technique has become a well-established imaging modality for the study of such activity, providing good spatial resolution and allowing reliable quantification of dopaminergic activity, ultimately identifying the different parts of the striatum and quantifying levels of activity, which makes it very useful for research purposes. The 18F-FDOPA PET and 99mTc-TRODAT-1 SPECT techniques differ with respect to the binding sites. Because 18F-FDOPA measures aromatic L-amino acid decarboxylase activity and 99mTc-TRODAT-1 measures DAT availability, each is susceptible to different compensatory mechanisms. In 99mTc-TRODAT-1 SPECT, compensation in early-stage PD consists in downregulation of presynaptic DATs in order to maintain high dopamine concentrations within the synapse. That increases the diagnostic sensitivity of this technique during the early stages of the disease. To our knowledge, there has been only one study demonstrating that 99mTc-TRODAT-1 SPECT has adequate sensitivity and specificity for the diagnosis of PD(7). One previous study compared 99mTc-TRODAT-1 SPECT and 18F-FDOPA PET in patients with PD in Taiwan(17). However, there have been no such studies employing 18F-FDOPA PET as the gold-standard for evaluating dopaminergic function and comparing it with 99mTc-TRODAT-1 SPECT in patients with PD in Latin America. In the present study, the semiquantitative analysis showed that the variation within ROIs was greater for 99mTc-TRODAT-1 SPECT than for 18F-FDOPA PET, likely because of the lower spatial resolution of SPECT, together with the nonspecific uptake of 99mTc-TRODAT-1 and the higher counts obtained with PET, which would result in significant variations in quantification. In one instance (the caudate nucleus in subject 5), there was not even agreement between the quantitative and qualitative analyses. Therefore, we believe quantitative analysis in 99mTc-TRODAT-1 SPECT should not be interpreted separately from a visual analysis by an experienced observer. Our study has some limitations. Pathology specimens were not available to confirm PD in the individuals recruited, and the diagnosis was therefore established on the basis of the clinical findings alone, in accordance with currently accepted criteria(1). In addition, because this was a pilot study, the number of subjects was limited. Nevertheless, the statistical power attained for the correlation analysis was acceptable. Comparative studies have shown that SPECT techniques involving the use of tracers such as iodine-123-ß-CIT can be good alternatives to PET(18,19). In 2011, the FDA approved the use of the DaTscan technique for the study of dopaminergic function, and the technique is now widely used. In contrast, the use of 99mTc-TRODAT-1 SPECT has yet to be approved by the FDA or the EMEA. Nevertheless, in many countries, where 99mTc-labeled tracers are more accessible and less expensive, 99mTc-TRODAT-1 SPECT has become a widely available alternative in clinical practice. CONCLUSION Given the accessibility and lower cost of 99mTc-TRODAT-1 SPECT, together with the fact that it appears to correlate well with 18F-FDOPA PET, we believe that it should be considered a viable option for the imaging of patients with PD in daily practice. REFERENCES 1. Berg D, Adler CH, Bloem BR, et al. Movement disorder society criteria for clinically established early Parkinsons disease. Mov Disord. 2018;33:16436. 2. Arena JE, Stoessl AJ. Optimizing diagnosis in Parkinsons disease: radionuclide imaging. Parkinsonism Relat Disord. 2016;22 Suppl 1: S4751. 3. Oravivattanakul S, Benchaya L, Wu G, et al. Dopamine transporter (DaT) scan utilization in a movement disorder center. Mov Disord Clin Pract. 2015;3:315. 4. Ibrahim N, Kusmirek J, Struck AF, et al. The sensitivity and specificity of F-DOPA PET in a movement disorder clinic. Am J Nucl Med Mol Imaging. 2016;6:1029. 5. Jennings D, Siderowf A, Stern M, et al. Imaging prodromal Parkinson disease: the Parkinson Associated Risk Syndrome Study. Neurology. 2014;83:173946. 6. Artzi M, Even-Sapir E, Shacham HL, et al. DaT-SPECT assessment depicts dopamine depletion among asymptomatic G2019S LRRK2 mutation carriers. PLoS One. 2017;12:e0175424. 7. Weng YH, Yen TC, Chen MC, et al. Sensitivity and specificity of 99mTc-TRODAT-1 SPECT imaging in differentiating patients with idiopathic Parkinsons disease from healthy subjects. J Nucl Med. 2004;45:393401. 8. Mittal BR, Sood A, Shukla J, et al. 99mTc-TRODAT-1 SPECT/CT imaging as a complementary biomarker in the diagnosis of parkinsonian syndromes. Nucl Med Commun. 2018;39:3128. 9. Shih MC, Franco de Andrade LA, Amaro Jr E, et al. Higher nigrostriatal dopamine neuron loss in early than late onset Parkinsons disease?a [99mTc]-TRODAT-1 SPECT study. Mov Disord. 2007;22:8636. 10. Nirenberg MJ, Vaughan RA, Uhl GR, et al. The dopamine transporter is localized to dendritic and axonal plasma membranes of nigrostriatal dopaminergic neurons. J Neurosci. 1996;16:43647. 11. Mozley PD, Schneider JS, Acton PD, et al. Binding of [99mTc]TRODAT-1 to dopamine transporters in patients with Parkinsons disease and in healthy volunteers. J Nucl Med. 2000;41:5849. 12. Huang WS, Lin SZ, Lin JC, at al. Evaluation of early-stage Parkinsons disease with 99mTc-TRODAT-1 imaging. J Nucl Med. 2001; 42:13038. 13. Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17:42742. 14. Goetz CG, Tilley BC, Shaftman SR, et al. Movement Disorder Society-sponsored revision of the Unified Parkinsons Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23:212970. 15. Chang LT. A method for attenuation correction in radionuclide computed tomography. IEEE Transactions on Nuclear Science. 1978; 25:63843. 16. García-Gómez FJ, García-Solís D, Luis-Simón FJ, et al. Elaboration of the SPM template for the standardization of SPECT images with 123I-ioflupane. Rev Esp Med Nucl Imagen Mol. 2013;32:3506. 17. Huang WS, Chiang YH, Lin JC, et al. Crossover study of (99m)Tc-TRODAT-1 SPECT and (18)F-FDOPA PET in Parkinsons disease patients. J Nucl Med. 2003;44:9991005. 18. Ishikawa T, Dhawan V, Kazumata K, et al. Comparative nigrostriatal dopaminergic imaging with iodine-123-beta CIT-FP/SPECT and fluorine-18-FDOPA/PET. J Nucl Med. 1996;37:17605. 19. Eshuis SA, Jager PL, Maguire RP, et al. Direct comparison of FP-CIT SPECT and F-DOPA PET in patients with Parkinsons disease and healthy controls. Eur J Nucl Med Mol Imaging. 2009;36:45462. 1. Fleni, Buenos Aires, Argentina 2. Pontificia Universidad Catolica Argentina, Buenos Aires, Argentina a. https://orcid.org/0000-0001-7044-1520 b. https://orcid.org/0000-0002-6894-4249 c. https://orcid.org/0000-0002-9703-7998 d. https://orcid.org/0000-0002-6230-3010 e. https://orcid.org/0000-0003-0155-901X f. https://orcid.org/0000-0003-3383-8566 g. https://orcid.org/0000-0001-6898-7823 Correspondence: Dr. Marcelo Merello Fleni. Montañeses 2325, C1428AQK Ciudad Autónoma de Buenos Aires, Argentina Email: mmerello@ fleni.org.ar Received 22 June 2020 Accepted after revision 4 August 2020 |
|
Av. Paulista, 37 - 7° andar - Conj. 71 - CEP 01311-902 - São Paulo - SP - Brazil - Phone: (11) 3372-4544 - Fax: (11) 3372-4554