 |
|
Antonio Rahal Junior1; Priscila Mina Falsarella1; Vinicius Tadeu Rodrigues Ferreira2; Guilherme Cayres Mariotti1; Marcos Roberto Gomes de Queiroz1; Rodrigo Gobbo Garcia1
RESUMO
OBJETIVO: Descrever a técnica de injeção de matrizes hemostáticas e a experiência do nosso serviço de radiologia intervencionista na sua aplicação. MATERIAIS E MÉTODOS: Foi realizado estudo unicêntrico com análise retrospectiva da experiência do nosso grupo na utilização de matriz hemostática gelatinosa em biópsias percutâneas. RESULTADOS: Foram realizadas 73 biópsias com utilização de matriz hemostática gelatinosa no trajeto da agulha coaxial em diferentes órgãos, como fígado, rim, baço, entre outros. A única complicação observada foi a migração da matriz hemostática para o sistema coletor do rim esquerdo, sendo solucionada com tratamento clínico. Não foram observados casos de sangramento no trajeto das biópsias após a injeção de matrizes hemostáticas. CONCLUSÃO: O uso de matrizes hemostáticas no trajeto de biópsias percutâneas é mais uma ferramenta disponível a ser considerada nos procedimentos minimamente invasivos.
Palavras-chave: Técnicas hemostáticas; Hemostáticos/administração & dosagem; Biópsia por agulha/métodos; Hemorragia/prevenção & controle.
ABSTRACT
OBJECTIVE: To describe the technique of injecting hemostatic matrix, as well as the experience of our interventional radiology department in its application. MATERIALS AND METHODS: We conducted a single-center study with retrospective analysis of the experience of our group in the use of hemostatic gelatin matrix in percutaneous biopsies. RESULTS: In a total of 73 biopsies in different organs, such as the liver, kidney, and spleen, hemostatic gelatin matrix was introduced into the coaxial needle. The only complication observed was migration of the hemostatic matrix to the left kidney collecting system, and that was resolved with clinical treatment. There were no cases of bleeding after the injection of hemostatic matrix. CONCLUSION: The use of hemostatic matrices in the path of percutaneous biopsies is another tool available for consideration in minimally invasive procedures.
Keywords: Hemostatic techniques; Hemostatics/administration & dosage; Biopsy, needle/methods; Hemorrhage/prevention & control.
INTRODUÇÃO
Biópsias percutâneas são procedimentos seguros e pouco invasivos, quando comparados com as técnicas cirúrgicas convencionais, e são utilizados para diagnóstico histológico de lesões em órgãos sólidos(1). A despeito de se tratar de método seguro, algumas vezes tais procedimentos podem cursar com sangramento de difícil controle, sobretudo em pacientes com distúrbio de coagulação(2). Todavia, a não realização do procedimento pode retardar o início do tratamento apropriado.
O risco de sangramento de uma biópsia depende de vários fatores, que devem ser minuciosamente avaliados antes do procedimento(2). Distúrbio de coagulação é o fator que mais contribui no aumento do risco de sangramento(3), e os pacientes com quantidade de plaquetas sérica inferior a 50.000/mm3 e/ou razão normalizada internacional superior a 1,5 são os que apresentam risco aumentado para sangramento. Em oposição, há fatores que contribuem para a redução de sangramento, como a transfusão de hemoderivados, de acordo com a indicação e a suspensão de antiagregantes plaquetários ou anticoagulantes quando possível. No campo dos fatores técnicos, o planejamento do melhor acesso para atingir a lesão, com escolha do melhor método de imagem para orientação – seja ultrassonografia, tomografia, fusão de imagens de ultrassonografia com outras técnicas, tais como ressonância, tomografia e tomografia por emissão de pósitrons –, colabora para redução de risco de sangramento e maior precisão da técnica. O uso de agulha coaxial como guia para a agulha de corte também é elemento importante que reduz o risco de sangramento, uma vez que evita a repetição de trauma perfurocortante no trajeto da biópsia(4), além do uso da agulha de corte semiautomática, pela possibilidade de maior controle na progressão da sua extremidade.
Uma forma complementar de agregar segurança ao procedimento, reduzindo o risco de sangramento, é a realização da embolização do trajeto da agulha com matrizes hemostáticas, agentes já utilizados há algumas décadas em cirurgias abertas(5), mas de aplicação relativamente recente em procedimentos percutâneos guiados por imagem. Atualmente, várias matrizes hemostáticas são disponíveis para utilização, por exemplo, de gelatina, colágeno, celulose, e outras. Entre elas, a matriz de gelatina absorvível é a mais comumente utilizada(2). Visto que muitos dos pacientes que necessitam de biópsia são portadores de doenças com tendências hemorrágicas(6), como os hepatopatas e nefropatas(7), e considerando o risco aumentado para sangramento de grande parte das lesões focais, decorrente de sua hipervascularização, o conhecimento e a aplicação desta técnica, para casos selecionados, vem sendo de grande valia na prática médica, com especial enfoque na intervenção.
O objetivo deste estudo é descrever a técnica de injeção de matrizes hemostáticas, assim como a experiência do nosso serviço na sua aplicação, as complicações associadas e seu manejo pelo radiologista intervencionista.
MATERIAIS E MÉTODOS
Este é um estudo unicêntrico, com análise retrospectiva da experiência do nosso grupo na utilização de matriz hemostática gelatinosa à base de pele suína purificada (Gelfoam®; Pfizer, New York, NY, EUA) em biópsias percutâneas, realizado no centro de medicina intervencionista da nossa instituição.
Pacientes
No período de outubro de 2013 a maio de 2016, 73 pacientes (39 homens e 34 mulheres) com idade entre 12 e 84 anos (média: 54,1 anos; mediana: 54 anos) foram encaminhados ao setor de radiologia intervencionista do nosso hospital e submetidos a biópsia percutânea guiada por imagem com injeção de Gelfoam no trajeto da biópsia. Os critérios de inclusão foram pacientes com risco aumentado de sangramento por trombocitopenia ou coagulopatia (contagem de plaquetas inferior a 75.000/mm3 e/ou razão normalizada internacional superior a 1.5), ou pacientes nos quais, por terem comorbidades associadas, o risco de sangramento não seria aceitável. Os critérios de exclusão foram pacientes com contraindicação ao uso de Gelfoam por alergia conhecida a colágeno porcino. Todos os pacientes assinaram termo de consentimento informado antes da realização da biópsia.
Procedimento
Todas as biópsias foram realizadas com técnica asséptica, após planejamento do melhor trajeto, evitando-se estruturas nobres ou vasos, e guiadas por tomografia computadorizada (Somatom Definition AS 40-slice – Siemens, Berlim, Alemanha) ou por ultrassonografia com ou sem fusão de imagens (IU 22 Philips – Philips Healthcare, Andover, MA, EUA; Aplio 500 Platinum – Toshiba American Medical Systems, Tustin, CA, EUA; Logiq E9 VNav – General Electric Healthcare, Milwaukee, WI, EUA). Cada biópsia foi realizada por um entre dez experientes radiologistas intervencionistas (experiência de mais de cinco anos). A anestesia utilizada (anestésico local com ou sem sedação, ou anestesia geral associado a infiltração com anestésico local) variou de acordo com as características do local a ser biopsiado, posicionamento necessário e as características do paciente.
Os procedimentos foram realizados necessariamente com agulha coaxial 16, 17 ou 19 gauge, a depender da estrutura alvo, em associação com agulha de corte semiautomática 18 ou 20 gauge.
Técnica
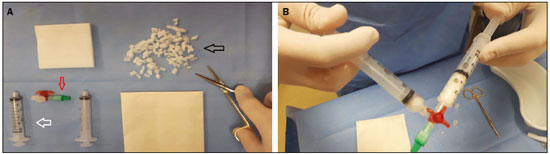
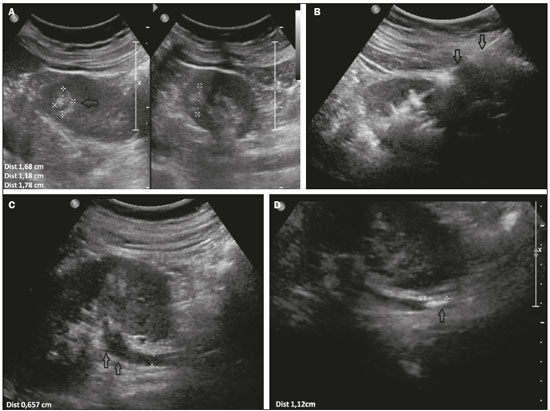
Finalizada a biópsia, Gelfoam cortado em pequenos pedaços de aproximadamente 2 a 3 mm ou em pó era misturado em soro fisiológico, utilizando-se duas seringas de 10 mL acopladas por uma torneira de três vias até a formação de uma pasta (Figura 1). O volume administrado foi cerca de 2 a 4 mL da pasta em cada caso, por meio de agulha coaxial, no trajeto da biópsia, iniciando-se no local alvo da biópsia até a cápsula do órgão, guiado por imagem.