Radiologia Brasileira - Publicação Científica Oficial do Colégio Brasileiro de Radiologia
AMB - Associação Médica Brasileira CNA - Comissão Nacional de Acreditação
 Vol. 48 nº 2 - Mar. / Apr. of 2015
Vol. 48 nº 2 - Mar. / Apr. of 2015
|
ORIGINAL ARTICLE
|
|
Multidetector computed tomography in the preoperative staging of gastric adenocarcinoma |
|
|
Autho(rs): Ricardo Hoelz de Oliveira Barros1; Thiago José Penachim1; Daniel Lahan Martins1; Nelson Adami Andreollo2; Nelson Marcio Gomes Caserta3 |
|
|
Keywords: Gastric cancer; Computed tomography; Staging. |
|
|
Abstract: INTRODUCTION
Gastric cancer is currently the fourth most common neoplasm worldwide, despite reports about decrease in the incidence in developed countries. Mortality rates are still high, and gastric cancer remains as the second cause of cancer-related deaths after lung cancer(1). Adenocarcinoma is the most frequent histological subtype of gastric cancer, representing about 95% of cases, and the other subtypes comprise lymphomas, tumors of stromal origin and other more rare subtypes as neuroendocrine tumors. Patients' survival is related to the tumor invasion depth and lymph node involvement. Five-year survival for patients with advanced tumors range between 7% and 27%, while five-year survival for patients with early-stage tumors achieves 85-100%(2). Some early-stage tumors (T1) may be endoscopically (mucosectomy) or laparoscopically resected. On the other hand, some protocols indicate neoadjuvant chemotherapy and/or radiotherapy in cases of advanced gastric cancer(3). Thus, the definition of an appropriate therapy depends on an accurate preoperative staging and may increase the cure rates and improve the patients' quality of life. The TNM (tumor/node/metastasis) is the most frequently utilized cancer staging system, and is currently at its seventh edition(4,5). Preoperative staging is frequently performed with abdominal ultrasonography, computed tomography and endoscopic ultrasonography(6). Until recently, endoscopic radiography was regarded as the best method of preoperative staging to determine the degree of tumor invasion (category T)(7). The most recent international consensus corroborated the necessity of preoperative TNM staging and pointed out multidetector-row computed tomography as the best staging method(8,9), which has demonstrated similar or superior accuracy as compared with endoscopic ultrasonography for T-staging and a clear advantage in relation to other methods for N- and M-staging. Multidetector-row computed tomography, particularly those apparatuses with 16 or more channels, offers rapid acquisition of submillimetric sections, isotropic multiplanar reconstruction and postprocessing options such as virtual endoscopy, which increases the method accuracy in the local staging(10-13). Additionally, computed tomography can evaluate lymph nodes and other organs(14). A significant advantage of magnetic resonance imaging over computed tomography in gastric cancer staging is still to be demonstrated(15-17). Few studies in the literature quantify the diagnostic performance of 64-detector-row computed tomography in the preoperative stating of gastric cancer, the Brazilian literature concerning this topic is particularly scarce. The present study was aimed at evaluating the role played by multidetector-row computed tomography in the local preoperative staging of gastric adenocarcinoma, determining sensitivity, specificity and accuracy of the method for T, N, and M, as well as the global accuracy for each of these categories. MATERIALS AND METHODS In the period between June 2009 and July 2011, 54 patients with endoscopic biopsy-proven gastric adenocarcinoma who had not yet been treated with surgery, chemotherapy or radiotherapy, where prospectively and preoperatively evaluated by multidetector-row computed tomography. Four out of those 54 patients were excluded for not having been submitted to laparotomy since all of them presented with metastatic disease and with no need for palliative surgery at the moment of their initial clinical evaluation. Among the remaining 50 patients, 22 (44%) were women, and 28 (56%) were men. At the moment of the surgery the patients' age ranged between 33 and 88 years (mean age 61.6 years). In the present study, only cases of adenocarcinomas were considered, and other neoplastic lesions, such as lymphomas were excluded. Computed tomography protocol Computed tomography scans were performed in a 64-detector row computed tomography apparatus (Aquillion 64; Toshiba Medical Systems, Tochigi, Japan), in patients after minimum 8-hour fasting. The patients were informed about the procedure and signed a term of free and informed consent. Initially, the patients ingest 750 mL to 1000 mL of water over 10 to 15 minutes as a means to achieve appropriate gastric distension. Then, a volumetric acquisition of the upper abdomen was performed before the intravenous iodinated contrast injection. All the patients were scanned in supine position. Subsequently, volumetric acquisitions were performed as follows: upper abdomen at arterial phase; total abdomen at portal phase (40 seconds after the arterial phase); and, again, upper abdomen at the equilibrium phase, 150 seconds after the arterial phase. The detection start was automatically triggered by the apparatus after appropriate contrast enhancement of the arterial vascular system (SureStart®; Toshiba Medical Systems, Japan). A non-ionic venous contrast agent (Iopromide - Ultravist 300; Bayer Schering Pharma AG, Berlin, Germany), at 1.5 mL/kg injected into the cubital vein through a 18G or 20G venous catheter, and flow of 3.5 mL/s, by means of an automated infusion pump (MedRad; Warrendale, USA). The non-contrast-enhanced phase was utilized for detecting liver steatosis and pathological calcifications, and as a reference for comparison with de enhancement pattern of eventually detected gastric, lymph node or metastatic lesions. The arterial and delayed phases were utilized to evaluate the level tumor invasion depth (T staging) and to characterize eventual liver lesions according to the protocol described by Chen et al.(12), and the portal phase was utilized to evaluate lymph nodes (N staging) and other abdominal structures (M staging). The utilized technical tomographic parameters were the following: slice thickness 64 × 0.625 mm, reconstruction thickness 1 mm, computed tomoghraphy scanner rotation speed 0.5 s, 120 kVp and automatic tube current modulation (mAs), aimed at optimizing and reducing the effective radiation dose. After acquisition, the images were stored as DICOM files on DVD-type media for later analysis on a radiological workstation. Staging definitions and images analysis Two radiologists specialized in abdominal diseases, both with 7-year experience in abdominal radiology, were responsible for the images analysis. They were blinded to the endoscopic results, and to lesions size and sites. The images were analyzed with the Osirix Imaging software specialized in DICOM images processing (Pixmeo; Genève, Switzerland). Then, the tomographic staging data were compared with the obtained surgical and anatomopathological results. By the time of the present study design, implementation and data collection, the sixth edition of the TMN Staging System was effective. Later in 2010, the seventh edition was published and the authors reclassified the data according to the new classification, specifying the respective staging categories, as necessary. The definitions utilized in the tomographic and pathological staging are described on Table 1. At computed tomography, lymph nodes were considered positive as they measured > 6 mm in their smallest axis and located in perigastric chains or as they measured > 8 mm in their smallest axis and located in the other chains. Also, clusters of three or more lymph nodes in a single chain were considered positive, even in cases of normal-sized lymph nodes. Lymph nodes were grouped into categories according to seventh edition of the TNM Staging System, as follows: N0 - no lymph node disease; N1 - 1 or 2 positive lymph nodes; N2 - 3 to 6 positive lymph nodes; N4 - 7 or more positive lymph nodes. Only the number of radiologically positive perigastric lymph nodes was considered, without considering the affected chain or the distance in relation to the gastric tumor, according to the seventh edition of the TNM Staging System. Finally, the patients were classified either as M0 or M1, according to the absence or presence of metastatic disease signs. Statistical analyses were performed to obtain values for sensitivity, specificity and accuracy in the staging of gastric adenocarcinomas for T, N and M categories, as well as the global accuracy for each category. The interobserver agreement was evaluated with the weighted Kappa method, according to Landis and Koch (< 0: poor agreement; 0.01-0.20: slight agreement; 0.21-0.40: fair agreement; 0.41-0.60: moderate agreement; 0.61-0.80: substantial agreement; 0.81-1.0: almost perfect agreement). RESULTS Tumor invasion staging (T) The results regarding gastric cancer T staging are summarized on Table 2. The accuracy in the classification of T categories ranged between 74% and 96% for observer 1, and between 80% and 92% for observer 2. The global accuracy was 70% for both observers. 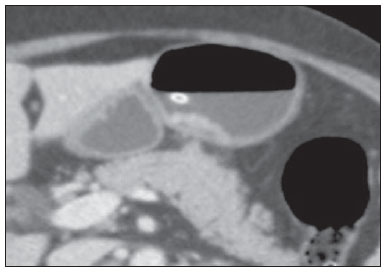 Figure 1. Focal thickening or abnormal enhancement of the gastric mucosa, with a preserved hypodense strip deep to the lesion, corresponding to the submucosal layer - category T1 at the histopathological analysis. 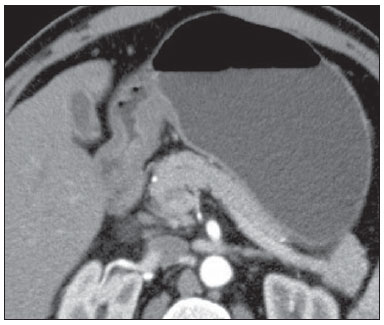 Figure 2. Thickening and abnormal enhancement involving the whole thickness of the gastric wall, with smooth external contour of the stomach - category T2 after histopathological analysis. 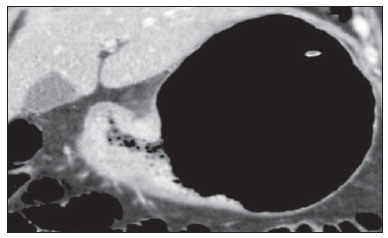 Figure 3. Thickening and abnormal enhancement involving the whole thickness of the gastric wall, associated with linear or reticular striations extending towards the perigastric fat - category T4A after histopathological analysis. 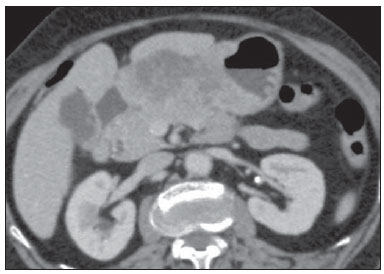 Figure 4. Signs of invasion of the pancreatic head and body - category T4B. The interobserver agreement was substantial, demonstrated by a weighted kappa value of 0.75. Then, the data were organized as to answer three questions of fundamental clinical significance in the preoperative staging: 1. Is the tumor in question an early-stage gastric cancer (T1 vs. T2-4)? Interobserver sensitivity ranged between 60% and 70%; specificity, between 95% and 98%; and accuracy, between 88% and 92%. 2. Does the tumor present extraserosal extension (T1-3 vs. T4)? Interobserver sensitivity ranged between 85% and 96%; specificity, between 74% and 83%; and accuracy, between 82% and 86%. 3. Does the tumor present extensive adjacent organs invasion (T1-4A vs. T4B)? Interobserver sensitivity ranged between 85% and 92%; specificity, between 84% and 100%; and accuracy, between 86% and 96%. Staging of lymph node involvement (N) The results regarding gastric cancer N staging are summarized on Table 3. 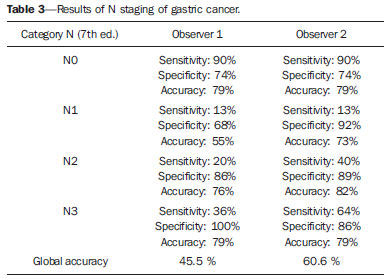 The accuracy in N staging ranged between 55% and 79% for observer 1, and between 73% and 82% for observer 2. The global accuracy was 45.5% for observer 1, and 60.6% for observer 2. Metastatic involvement staging (M) The results regarding gastric cancer M staging are summarized on Table 4. The global accuracy was 89.6% for both observers. 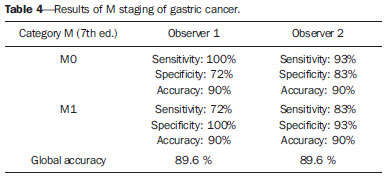 Three cases were classified as M0 at the preoperative evaluation, but peritoneal metastatic implants were intraoperatively found and were later histopathologically confirmed. After retrospective analysis, one of these cases presented a small amount of ascites in the posterior cul de sac, but the presence of peritoneal implant could not be tomographically detected in any of the cases. DISCUSSION In the present study, computed tomography demonstrated high accuracy in the T staging classification, particularly as regards its capacity to answer the more clinically relevant questions, namely: 1) whether the tumor in question is an early-stage gastric tumor (accuracy 88-92%); 2) whether, in the case of locally advanced tumors, there are signs of extraserosal extension (accuracy 82-86%); and, in case of a positive answer, 3) whether there are signs of adjacent organs invasion (accuracy 86-96%). The tomographic staging accuracy is lower in the differentiation between categories T2 and T3 (both restricted to the gastric wall), but such a differentiation is not critical in the preoperative investigation since the management is the same for both categories. The accuracy reported in previous studies for tumor invasion staging (T) ranges between 66% and 88%(10,16,18-22). In the present study, the authors found a global accuracy of 70% for both observers, in agreement with data reported by previous studies. Some factors have contributed to reduce the accuracy indices in the present study. A great proportion of advanced tumors was observed in the present study, in agreement with population data usually found in studies developed in Western countries, particularly in developing countries. In studies developed in Eastern countries, cases of early gastric cancer are more frequently reported. While in the present study 20% of the cases were staged as T1, Yang et al.(22), for example, reported histopathological staging T1 in 40% of the patients in their study sample. Considering that, in the T staging, the accuracy is higher for category T1, these clinical data reduce the global accuracy of the staging. Despite the absence of a clear evidence in the literature, the new (seventh) edition of the TNM Staging System will lead to lower accuracy indices in the tomographic staging, since all the cases of disease restricted to the gastric wall are divided into three categories (T1, T2 or T3) and only the cases with extraserosal extension are classified as T4, making such stratification more difficult for the radiologist. Most studies available in the literature were based on the sixth edition of the TNM Staging System, classifying tumors with extraserosal extension as T3. Currently, there are ongoing studies for clinical validation of the seventh edition of the TNM Staging System, and presently available evidences indicate that this seventh edition is better than the sixth edition for category T, but is worse for category N(23,24). Further clinical studies are still required to validate the preoperative staging based on the seventh edition. Another difficulty already reported by previous studies in the literature is that the presence of subtle reticular opacities in the perigastric fat adjacent to the tumor, which most frequently represents extraserosal tumor extension (category T4A) but occasionally result from inflammatory process, leads to false positive results when interpreted as tumor extension(25,26). In the present study, the authors found three cases of T3 interpreted as T4A (false-positive) by both observers. On the other hand, in the evaluation of lymph node involvement, also in agreement with the available literature(14,27,28), the results were not so good, with global accuracy of 45.5% for observer 1, and 60.6% for observer 2. However, computed tomography demonstrated a better result in category N0, with 90% sensitivity, 74% specificity, and 79% accuracy. Thus, the tomographic method could demonstrate the absence of secondary lymph node involvement with good accuracy level. There is no consensus in the literature about the lymph node size to be considered positive at computed tomography. The criteria adopted in the present study (> 6 mm in the shortest axis for perigastric lymph nodes, and > 8 mm for the other chains) result in higher sensitivity, but at the expense of a decrease in the findings specificity(27). Despite the clear correlation between lymph node dimensions and neoplastic involvement, computed tomography presents an inherent limitation, since lymph nodes enlargement may be reactional, inflammatory, and normal-sized lymph nodes may contain metastatic implants. The recent fusion of the computed tomography technology with positron emission tomography apparatuses may improve the lymph node staging specificity, but further studies are still necessary to validate its effectiveness. Computed tomography has also demonstrated excellent performance in the definition of metastatic disease with 89.6% accuracy. Metastatic involvement may occur by hematogenous, lymphatic or peritoneal spread. Currently, the greatest limitation of computed tomography is related to the detection of secondary peritoneal involvement(29). In the present study, three cases were classified as M0 in the preoperative staging, but presented small secondary peritoneal implants, which were confirmed after histopathological analysis. A retrospective re-evaluation of computed tomography images of these three patients revealed that one of these three cases presented a small amount of ascites in the posterior pelvic cul de sac as the sole finding, and in none of these cases one could detect the presence of peritoneal implants, even retrospectively, indicating the method limitation for detecting millimetric implants. In selected cases, further investigation with PET/CT and magnetic resonance imaging may increase the sensitivity and specificity in the study of peritoneal implants. Other frequently employed strategy is the utilization of diagnostic videolaparoscopy at the beginning of the surgical approach to detect secondary peritoneal involvement. The finding of ascites at computed tomography should be taken into consideration in the setting of an advanced gastric tumor. According to Yajima et al.(30), the presence of ascites predicts peritoneal carcinomatosis with 51% sensitivity and 97% specificity. Exposure to ionizing radiation is one disadvantage of computed tomography. The protocol described in the literature for preoperative staging of gastric cancer, with multiple pre- and post-contrast acquisitions implies a relatively high exposure to radiation. It is important to consider that the population affected by gastric cancer is in a relatively advanced age range (mean 66 years in the present study) and presents with neoplasms associated with high mortality rates. Additionally, there is no study proving equivalent accuracy with less extensive protocols, so future studies could shed some light on this matter. A frequently utilized strategy is to reduce the number of tomographic phases in the postoperative follow up of these patients. CONCLUSION Multi-detector row computed tomography is currently essential in the preoperative assessment of patients with gastric cancer, and its utilization is already included in the recommendations of the most recent international consensus. The tomographic method presents excellent accuracy in the staging of tumor invasion depth (T) and in the staging of metastatic neoplastic disease (M). Despite the good accuracy in the staging of patients without lymph node disease (N0), the method presents limitations in the staging of lymph node involvement. REFERENCES 1. Ferlay J, Soerjomataram I, Ervik M, et al. Globocan 2012 v1.0, Cancer incidence and mortality worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer; 2013. [cited 2013 Dec 12]. Available from: http://globocan.iarc.fr. 2. Thrumurthy SG, Chaudry MA, Hochhauser D, et al. The diagnosis and management of gastric cancer. BMJ. 2013;347:f6367. 3. Blakely AM, Miner TJ. Surgical considerations in the treatment of gastric cancer. Gastroenterol Clin North Am. 2013;42:337-57. 4. Edge S, Byrd DR, Compton CC, et al. AJCC cancer staging handbook. 7th ed. New York: Springer-Verlag; 2010. 5. Lee MH, Choi D, Park MJ, et al. Gastric cancer: imaging and staging with MDCT based on the 7th AJCC guidelines. Abdom Imaging. 2012;37:531-40. 6. Park SR, Kim MJ, Ryu KW, et al. Prognostic value of preoperative clinical staging assessed by computed tomography in resectable gastric cancer patients: a viewpoint in the era of preoperative treatment. Ann Surg. 2010;251:428-35. 7. Furukawa K, Miyahara R, Itoh A, et al. Diagnosis of the invasion depth of gastric cancer using MDCT with virtual gastroscopy: comparison with staging with endoscopic ultrasound. AJR Am J Roentgenol. 2011;197:867-75. 8. Coburn N, Seevaratnam R, Paszat L, et al. Optimal management of gastric cancer: results from an international RAND/UCLA expert panel. Ann Surg. 2014;259:102-8. 9. Dixon M, Cardoso R, Tinmouth J, et al. What studies are appropriate and necessary for staging gastric adenocarcinoma? Results of an international RAND/UCLA expert panel. Gastric Cancer. 2014;17:377-82. 10. Kim AY, Kim HJ, Ha HK. Gastric cancer by multidetector row CT: preoperative staging. Abdom Imaging. 2005;30:465-72. 11. Kim YH, Lee KH, Park SH, et al. Staging of T3 and T4 gastric carcinoma with multidetector CT: added value of multiplanar reformations for prediction of adjacent organ invasion. Radiology. 2009;250:767-75. 12. Chen CY, Hsu JS, Wu DC, et al. Gastric cancer: preoperative local staging with 3D multi-detector row CT - correlation with surgical and histopathologic results. Radiology. 2007;242:472-82. 13. Moschetta M, Stabile Ianora AA, Anglani A, et al. Preoperative T staging of gastric carcinoma obtained by MDCT vessel probe reconstructions and correlations with histological findings. Eur Radiol. 2009;20:138-45. 14. Kwee RM, Kwee TC. Imaging in assessing lymph node status in gastric cancer. Gastric Cancer. 2009;12:6-22. 15. Kwee RM, Kwee TC. Imaging in local staging of gastric cancer: a systematic review. J Clin Oncol. 2007;25:2107-16. 16. Sohn KM, Lee JM, Lee SY, et al. Comparing MR imaging and CT in the staging of gastric carcinoma. AJR Am J Roentgenol. 2000;174:1551-7. 17. Motohara T, Semelka RC. MRI in staging of gastric cancer. Abdom Imaging. 2002;27:376-83. 18. Habermann CR, Weiss F, Riecken R, et al. Preoperative staging of gastric adenocarcinoma: comparison of helical CT and endoscopic US. Radiology. 2004;230:465-71. 19. Kumano S, Okada M, Shimono T, et al. T-staging of gastric cancer of air-filling multidetector-row CT: comparison with hydro-multidetector-row CT. Eur J Radiol. 2012;81:2953-60. 20. Bhandari S, Shim CS, Kim JH, et al. Usefulness of three-dimensional, multidetector row CT (virtual gastroscopy and multiplanar reconstruction) in the evaluation of gastric cancer: a comparison with conventional endoscopy, EUS, and histopathology. Gastrointest Endosc. 2004;59:619-26. 21. Shimizu K, Ito K, Matsunaga N, et al. Diagnosis of gastric cancer with MDCT using the water-filling method and multiplanar reconstruction: CT-histologic correlation. AJR Am J Roentgenol. 2005;185:1152-8. 22. Yang DM, Kim HC, Jin W, et al. 64 multidetector-row computed tomography for preoperative evaluation of gastric cancer: histological correlation. J Comput Assist Tomogr. 2007;31:98-103. 23. Graziosi L, Marino E, Cavazzoni E, et al. Prognostic value of the seventh AJCC/UICC TNM classification of non-cardia gastric cancer. World J Surg Oncol. 2013;11:103. 24. Hayashi T, Yoshikawa T, Bonam K, et al. The superiority of the seventh edition of the TNM classification depends on the overall survival of the patient cohort: comparative analysis of the sixth and seventh TNM editions in patients with gastric cancer from Japan and the United Kingdom. Cancer. 2013;119:1330-7. 25. Chen CY, Wu DC, Kang WY, et al. Staging of gastric cancer with 16-channel MDCT. Abdom Imaging. 2006;31:514-20. 26. Makino T, Fujiwara Y, Takiguchi S, et al. Preoperative T staging of gastric cancer by multi-detector row computed tomography. Surgery. 2011;149:672-9. 27. Mönig SP, Zirbes TK, Schröder W, et al. Staging of gastric cancer: correlation of lymph node size and metastatic infiltration. AJR Am J Roentgenol. 1999;173:365-7. 28. Morgagni P, Petrella E, Basile B, et al. Preoperative multidetectorrow computed tomography scan staging for lymphatic gastric cancer spread. World J Surg Oncol. 2012;10:1-5. 29. Kim SJ, Kim HH, Kim YH, et al. Peritoneal metastasis: detection with 16- or 64-detector row CT in patients undergoing surgery for gastric cancer. Radiology. 2009;253:407-15. 30. Yajima K, Kanda T, Ohashi M, et al. Clinical and diagnostic significance of preoperative computed tomography findings of ascites in patients with advanced gastric cancer. Am J Surg. 2006;192:185-90. 1. MDs., Radiologists at Hospital de Clínicas - Universidade Estadual de Campinas (Unicamp), Campinas, SP, Brazil 2. PhD, Full Professor, Department of Surgery - Universidade Estadual de Campinas (Unicamp), Campinas, SP, Brazil 3. PhD, Professor, Department of Radiology - Universidade Estadual de Campinas (Unicamp), Campinas, SP, Brazil Mailing Address: Dr. Ricardo Hoelz de Oliveira Barros Universidade Estadual de Campinas - Departamento de Radiologia Rua Tessália Vieira de Camargo, 126, Cidade Universitária Zeferino Vaz Campinas, SP, Brazil, 13083-970 E-mail: rhobarros@hotmail.com Received March 17, 2014. Accepted after revision October 2, 2014. Study developed at Department of Radiology - Universidade Estadual de Campinas (Unicamp), Campinas, SP, Brazil. |
|
Av. Paulista, 37 - 7° andar - Conj. 71 - CEP 01311-902 - São Paulo - SP - Brazil - Phone: (11) 3372-4544 - Fax: (11) 3372-4554